Lorem ipsum dolor sit amet, consectetur adipiscing elit. Praesent ut eros nec lectus varius sagittis. Integer non tortor quam. Aenean fringilla eu dolor eget varius. Phasellus augue est, tincidunt porta justo quis, tempor cursus erat. Vivamus elit lorem, egestas non...
GMP-Focused Pharmaceutical Manufacturing and Quality Services in Sweden
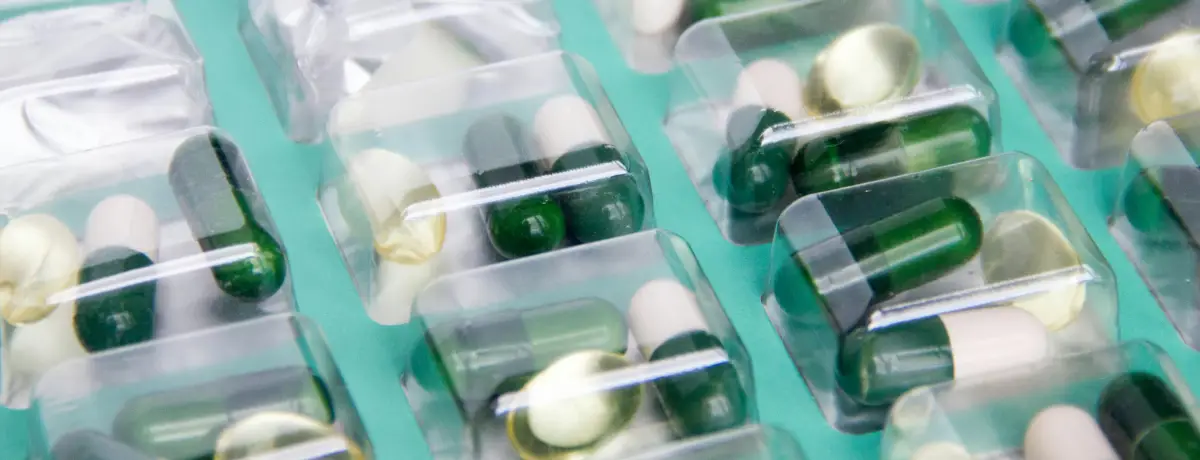
Pharmtronica AB is a Sweden based pharmaceutical company operating a GMP-focused site in Stockholm, with capabilities in contract manufacturing, patient-specific magistral (extemporaneous) preparations, import and handling of raw materials and APIs, secondary packaging and labelling, and quality control support.
We are Pharmtronica
Pharmtronica AB is part of the Chemtronica group and was established in 2024 to focus on pharmaceutical manufacturing and related services. At our GMP-focused site in Stockholm, Sweden, operations are supported by qualified persons, registered pharmacists, and GMP trained personnel across quality, production, and site functions.
We work according to defined procedures and a structured quality system, with a strong focus on product quality, patient safety, compliance, and traceability. Backed by the wider Chemtronica group, Pharmtronica combines specialised pharmaceutical expertise with the strength of an established business foundation.
Our Business and Service Areas
Three main business areas: Magistral Manufacture, Contract Manufacture, and API Supply. Through these services, we support healthcare and industry partners with quality-driven operations, trained personnel, and a structured approach to compliance, documentation, and traceability.
Tailored patient precision
Magistral Manufacture
We provide patient-specific magistral manufacture through controlled non-sterile preparation processes, supported by pharmacists, defined procedures, and quality review before release. Approved clients can access Pharmtronica’s designated ordering process for magistral services following registration with our customer service team.


Structured production quality
Contract Manufacture
We support contract manufacturing of selected pharmaceutical products, including liquid and semi-solid formulations, with structured processes for handling, packaging, labelling, documentation, and quality oversight.
Structured production quality
Contract Manufacture
We support contract manufacturing of selected pharmaceutical products, including liquid and semi-solid formulations, with structured processes for handling, packaging, labelling, documentation, and quality oversight.

Compliant ingredient management
API Supply
We support contract manufacturing of selected pharmaceutical products, including liquid and semi-solid formulations, with structured processes for handling, packaging, labelling, documentation, and quality oversight.

Vision, values and culture
At Pharmtronica AB, our vision is to contribute to safe and reliable pharmaceutical supply through quality-driven manufacturing and responsible operations. Our values are rooted in scientific rigour, GMP compliance, and respect for the responsibility that comes with working in pharmaceutical production.
We promote a culture where quality is part of everyday work, where trained professionals take clear ownership of their roles, and where continuous learning, traceability, and ethical collaboration support long-term trust with customers, healthcare partners, and authorities.

News
We now deliver to Norway
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Praesent ut eros nec lectus varius sagittis. Integer non tortor quam. Aenean fringilla eu dolor eget varius. Phasellus augue est, tincidunt porta justo quis, tempor cursus erat. Vivamus elit lorem, egestas non...
Global medical UESCA fair 2026
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Praesent ut eros nec lectus varius sagittis. Integer non tortor quam. Aenean fringilla eu dolor eget varius. Phasellus augue est, tincidunt porta justo quis, tempor cursus erat. Vivamus elit lorem, egestas non...
Get in touch
Get in touch with Pharmtronica AB for service enquiries, partnership discussions, or further information about our operations.